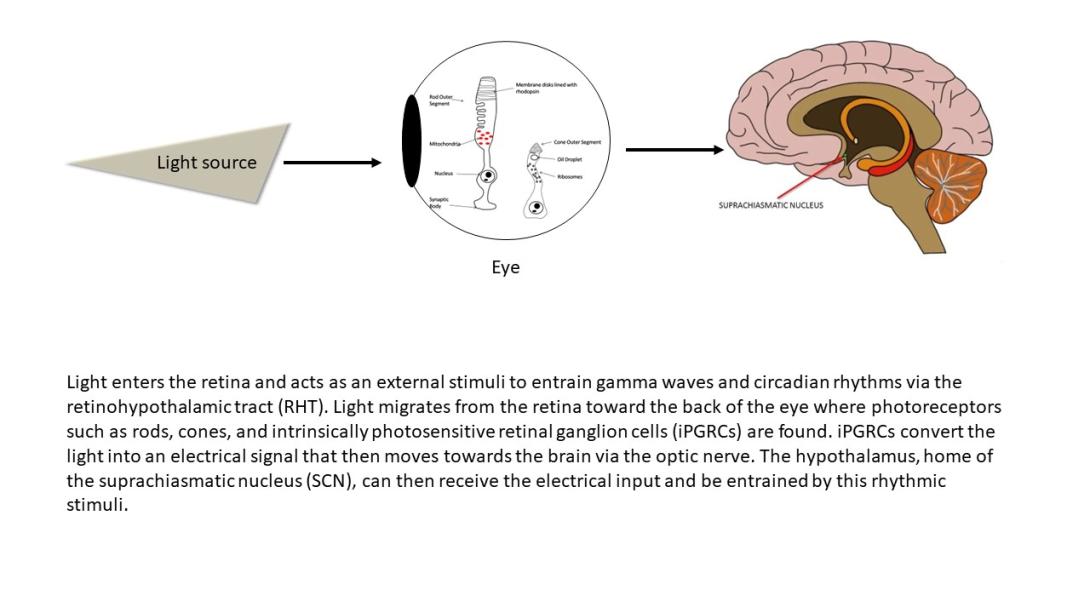
The brain is a network, and as we learn more about the pathophysiology of neurologic diseases, it is increasing clear that they are not isolated to a specific brain region but emerge from a dysfunction in brain networks. Many diseases are also a direct or indirect result of inflammatory events both in the brain and systemic. Indeed, epilepsy, Alzheimer's disease, Parkinson’s disease, neuropathic pain and depression all exhibit loss of neurons and reduced neuronal density as well as altered connectivity of key regions, and in many cases, the hallmark of inflammation is coincident with such neuronal death. As we begin to identify some of the circuits common to pathophysiology of many of these diseases, we can take advantage of new, minimally invasive, highly targeted interventions, such as neuromodulation, to help treat patients with debilitating symptoms but no cure. We are focusing on two quite distinct neuromodulatory pathways: (1) magnetogenetics and (2) control of circadian rhythms, both that can address potential therapeutic modalities in the future. However, our studies to date remain focused on gaining a mechanistic understanding of these two areas of neuromodulation.